How It Works: Aclarity PFAS Destruction Technology

Using Technology to Destroy PFAS, Forever.
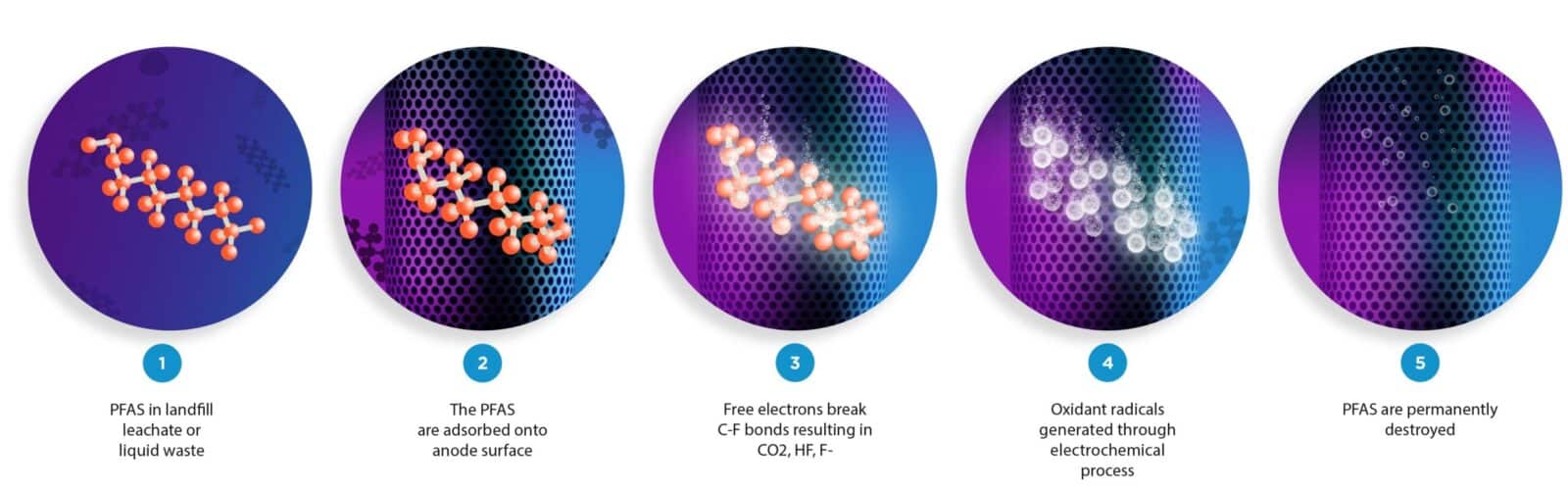
The Aclarity system destroys PFAS using a proprietary electrochemical process to break down contaminants. We work with treatment facilities, municipalities, landfills and other organizations to solve large, cost prohibitive water problems upstream. In drinking water, the contaminants are destroyed and clean water passes out the other side of the Aclarity system.
Forever Chemicals? No more.


Meeting regulations is just the beginning
How did a waste treatment facility in Michigan address high PFOA and PFOS levels and concerns around upcoming CERCLA regulations on hazardous substances? They sought out Aclarity for help. Read about this and other projects in the white paper "The Definitive Guide to PFAS Technology".
Addressing the Status Quo.
Aclarity is the first PFAS destruction technology in the world that is being deployed in a full-scale system. Other alternatives, such as removal and disposal, move the problem around, wasting more resources along the way. Aclarity’s technology has the ability to destroy PFAS forever, removing the majority of PFAS from our environment and preventing PFAS from migrating and contaminating even further.

Stay in the Loop
Sign up with your email address to receive news and updates.
